Press Releases
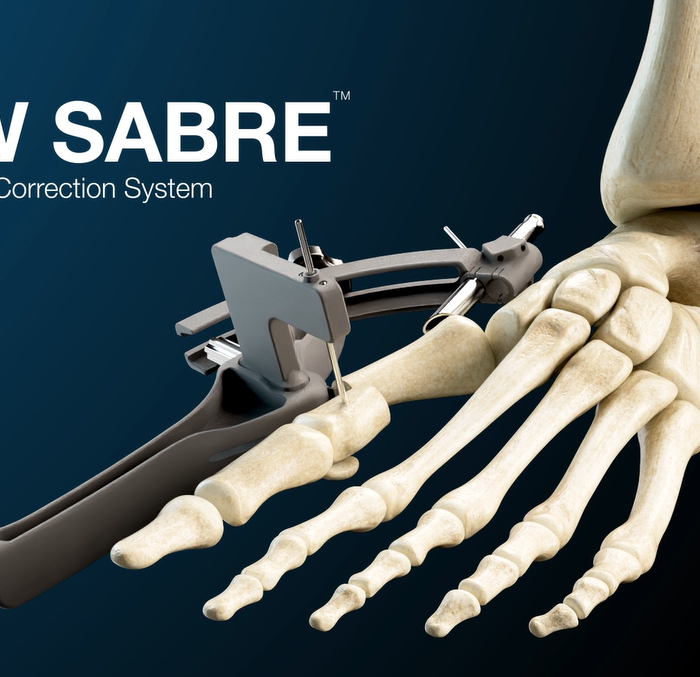
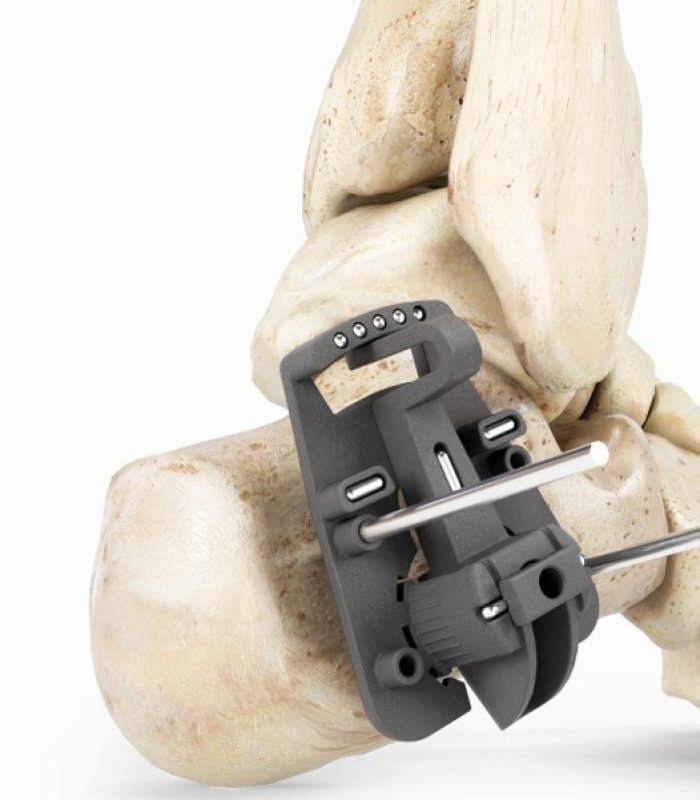
GLW Medical Continues To Expand Its MIS Solutions, Introduces GLW Sabre™ MIS Bunion System
Englewood Cliffs, NJ – GLW, Inc. (“GLW Medical” or the “Company”), a medical technology company focused on advancing orthopedic extremity and trauma surgery, today announced a preview of its forthcoming GLW Sabre™ (the “System”) MIS bunion correction system, an innovative, guided instrumentation and implant platform. The System allows for percutaneous keyhole incisions designed to reduce … Continued
GLW Medical to Expand MIS Solutions and Introduce Creed Edge™
Englewood Cliffs, NJ – GLW, Inc. (“GLW Medical” or the “Company”), a medical technology company focused on advancing orthopedic extremity and trauma surgery, today announced its upcoming product showcase at the 2025 American Orthopaedic Foot & Ankle Society (AOFAS) Annual Meeting, taking place September 10–13 in Savannah, Georgia. GLW Medical will debut the Creed Edge Cannulated … Continued

GLW Medical Announces Full Market Release of the Apollo Ankle Fracture Plating System
Englewood Cliffs, NJ., — GLW, Inc. (“GLW Medical” or the “Company”) with a focus on the foot and ankle orthopedic market is excited to announce the full market release of its Apollo Ankle Fracture (AFX) Plating System. The Company has commercialized its patented, first of its kind, additive manufactured titanium shell polyetheretherketone filled (TI-PEEK) plates … Continued
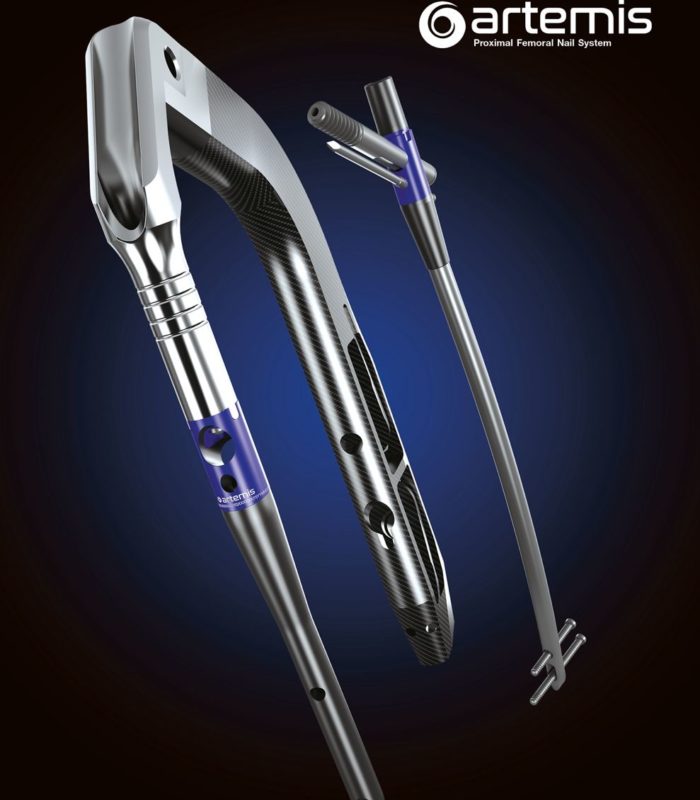
GLW Medical Announces Additional FDA 510(K) Clearance for the Artemis Proximal Femoral Nail System
NEW LONG NAIL TO STRENGTHEN THE ARTEMIS PFN SYSTEM AND STREAMLINE WORKFLOWS Englewood Cliffs, NJ – GLW, Inc. (“GLW Medical” or the “Company”), a medical technology company focused on advancing orthopedic extremity and trauma surgery, today announced additional U.S. Food and Drug Administration (FDA) 510(k) clearance of the Artemis Proximal Femoral Long Nail (PFN) System, … Continued

GLW Medical Receives FDA 510(k) Clearance for Apollo Ankle Fracture Plating System
Englewood Cliffs, NJ – GLW, Inc. (“GLW Medical” or the “Company”), a medical technology company focused on advancing orthopedic extremity and trauma surgery, today announced it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) to market its upcoming Apollo Ankle Fracture (AFX) Plating System, a novel portfolio of see-through, “ortholucent” bone … Continued

Solvay and GLW Medical Partner on Creed™ Cannulated Compression Screws for Orthopedics
Alpharetta, Ga., — Solvay’s Zeniva® polyetheretherketone (PEEK) resin was chosen by GLW, Inc (“GLW Medical”) for its new Creed™ Cannulated Screw System for foot and ankle surgery. This advanced new screw system recently received 510(k) clearance from the U.S. Food and Drug Administration (FDA). To create the new system’s ortholucent, plastic-metal hybrid composite, Solvay’s implant-grade radiolucent Zeniva® PEEK resin is … Continued

GLW Medical Announces FDA 510(K) Clearance for the Artemis Proximal Femoral Nail System
A FOURTH-GENERATION HIP FRACTURE NAIL MANUFACTURED USING A REVOLUTIONARY PROCESS Englewood Cliffs, NJ – GLW, Inc. (“GLW Medical” or the “Company”), a medical technology company focused on advancing orthopedic extremity and trauma surgery, today announced that it has received 510(k) clearance from the U.S. Food and Drug Administration for its Artemis Proximal Femoral Nail (PFN) … Continued
